Overview
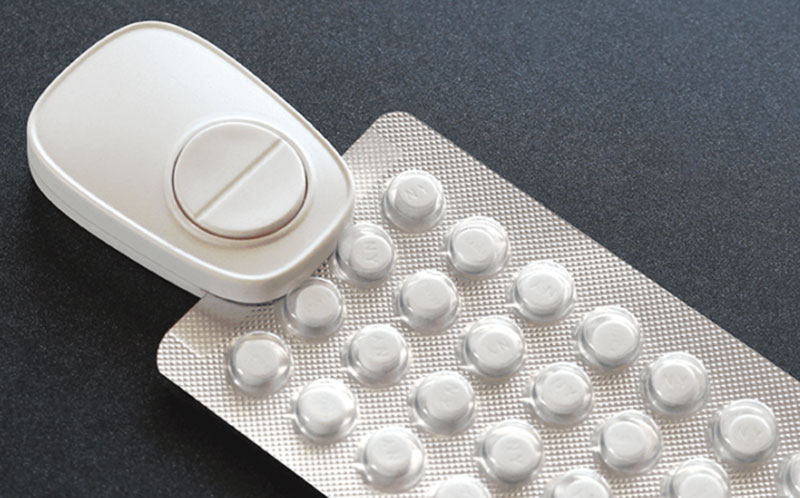
Popit is a Finnish startup that has developed a platform and sensing device. The company originally raised funds for the product in part via the crowdfunding site Indiegogo. Since then, the company has scaled up it’s offerings significantly, partnering with pharmaceutical companies such as Pfizer to improve patient adherence to their products. Popit was originally envisioned as a birth-control solution, but has since expanded to other medical areas.
How it works
The Popit sensor is a clip that attaches to a blister pack and links to a patient’s smartphone. When the patient expresses their pill from the pack, the sensor logs this information, keeping an electronic adherence record of pill-taking behavior. The platform provides reminders to patients if they miss a dose.
Scoring
Popit has been scored below according to it’s ability to serve the TB market for low- and middle-income countries (LMICs) along Fit for Purpose and Technical Specifications metrics. Note that higher scores equate to a greater ability to serve the market. For example, if a product scores well in the Provider Burden subcategory, that means that the burden for the provider is low.
For full descriptions of the sub-categories, see the 2021 DAT Market Landscape.
Fit for Purpose
Technical Specifications
Advantages
- Product is designed to work seamlessly with TB-type blister medication packs
- Sensor has a long battery life
- Device is extremely small and light-weight, enables automatic reporting, and has a high level of accuracy
Disadvantages
- Smartphones are required to use the product
- Popit has not been used extensively in LMICs or for TB
- To use with MDR TB, multiple devices would likely need to be purchased and used simultaneously
Access
To learn more, please visit the Popit website.